 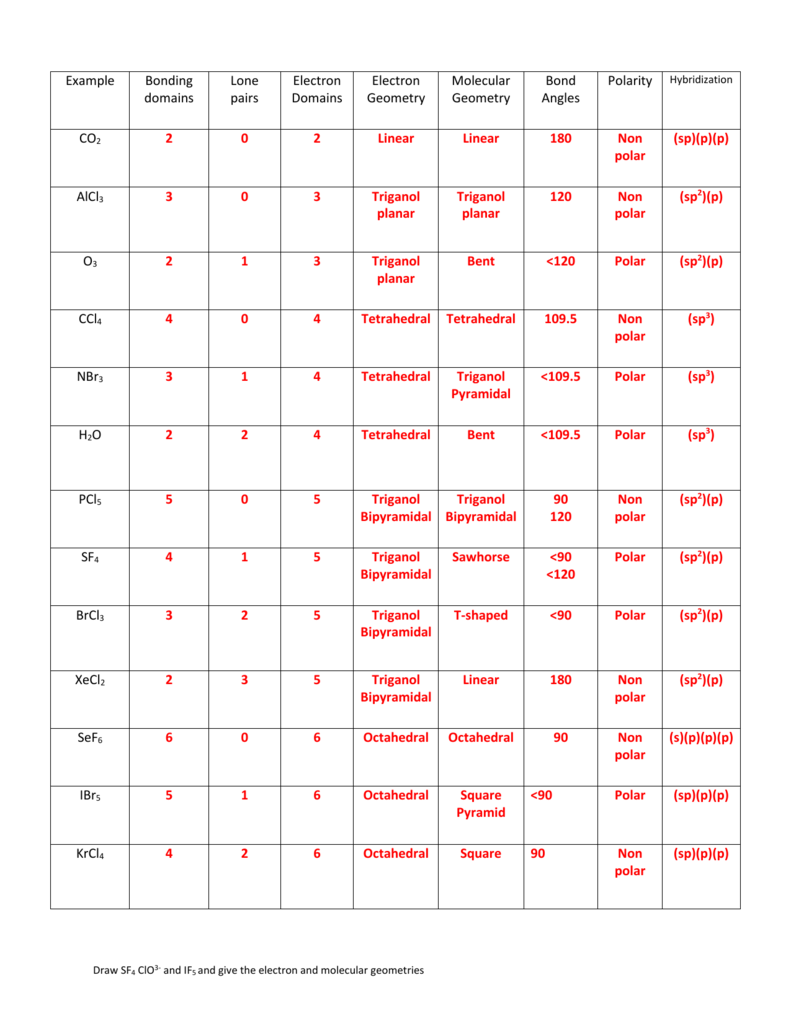
This shows us two double bonds around the carbon atom-each double bond counts as one electron group, and there are no lone pairs on the carbon atom. Let’s see how to determine the electron-pair geometry and molecular structure of CO 2 and BCl 3. The following examples illustrate the use of VSEPR theory to predict the molecular structures. 
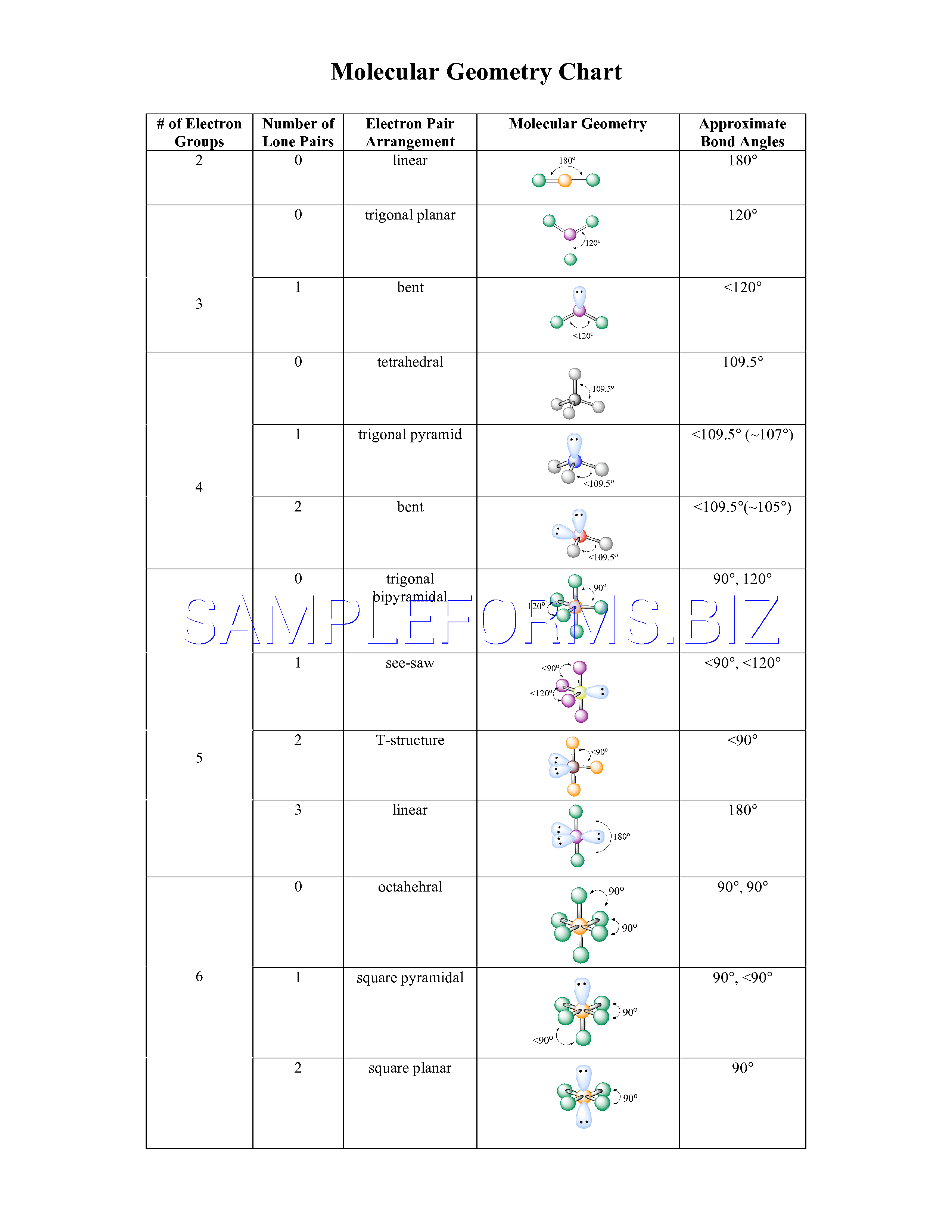
Predicting Molecular Structures using VSEPR Theory For a particular number of electron pairs, the molecular structures for one or more lone pairs are determined based on modifications of the corresponding electron-pair geometry. The molecular structures are identical to the electron-pair geometries when there are no lone pairs present. In an octahedral arrangement with two lone pairs, repulsion is minimized when the lone pairs are on opposite sides of the central atom. In trigonal bipyramidal arrangements, repulsion is minimized when every lone pair is in an equatorial position. If more than one arrangement of lone pairs and chemical bonds is possible, choose the one that will minimize repulsions, remembering that lone pairs occupy more space than multiple bonds, which occupy more space than single bonds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
